By Selin Yilmaz
This year, the Nobel Prize in Physiology and Medicine was awarded jointly to Mary E. Brunkow, Fred Ramsdell and Shimon Sakaguchi “for their discoveries concerning peripheral immune tolerance”. What exactly does this mean?

Our immune system is like our body’s army. It identifies foreign objects such as viruses, microbes and bacteria that may be harmful to us and keeps them under control, aiming to protect us. However, all pathogens are different, which complicates the immune system’s job. Some have even evolved to emulate body cells to bypass our immune system. If that’s so, how does the immune system recognise what is harmful and what isn’t? How does it avoid attacking our own cells?
Let’s continue our analogy. Some soldiers in this army are called T cells, of which there are multiple types. One of these is the Helper T Cells. These cells are support units, strengthening other immune cells and assisting in the activation of our antibody factories. Killer T cells, another type of T cell, destroy body cells that have already been infected by the invading pathogen, in an attempt to protect other cells.
All T cells have proteins on their surfaces known as T-cell receptors. Using these proteins, T cells interact with other cells and sense if the body is under attack. Similar to how pathogens all have different structures, these receptors, constructed from randomly combined genes, also have varied conformations. Theoretically, we can produce over 1015 different receptors! This allows our T-cells to detect many unique pathogens– how cool is that! However, with so many different T-cell receptors, wouldn’t some of them react to the body’s own cells as well?
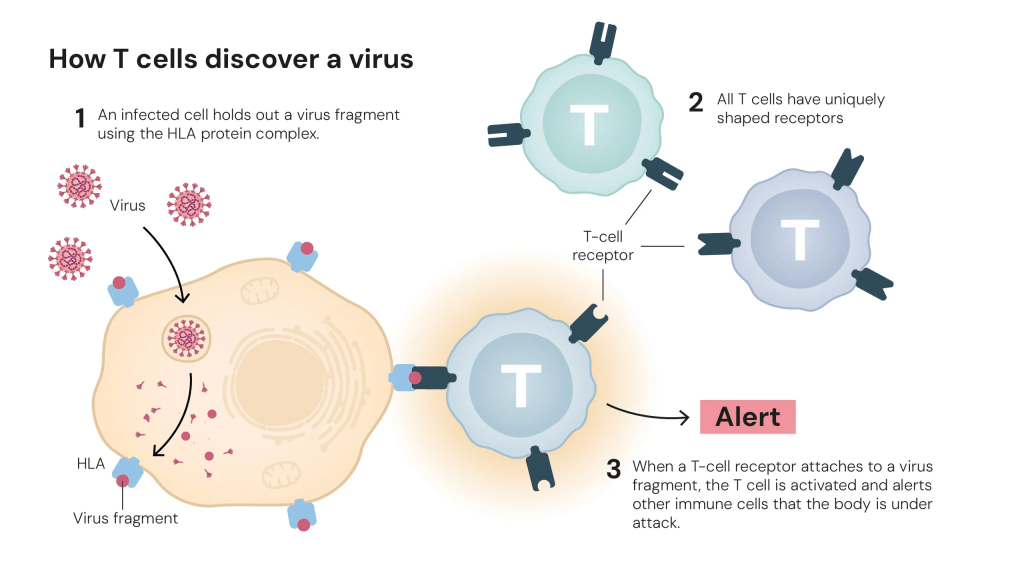
It was understood in the 1980s that T-cells mature in the thymus, a gland located in the upper chest. Here, T cells are subjected to a test that eliminates T-cells that react to the body’s own cells. This process is called the central tolerance. To understand the function of this organ in T-cell development, Shimon Sakaguchi and his colleagues surgically removed it from newborn mice. When the thymus was removed from three-day-old mice, they developed autoimmune diseases, meaning that the immune system was overactive and destroying the body. But when injected with mature T-cells from healthy mice, these diseases weren’t developed. This uncovered the existence of some type of T-cell that regulates other T cells from becoming overactive. After over a decade of working on the differentiation between different T-cells, Sakaguchi discovered a new type called regulatory T-cells. However, more proof was needed to cement this discovery.
In the 1940s, in a laboratory located in Tennessee, male mice were born with a mutation causing scaly and flaky skin, an enlarged spleen and lymph glands, and a shortened life span. In the 1990s, technology improved enough for researchers to begin exploring this mutation named scurfy. It was discovered that the mice’s organs were under attack by their own T-cells. At the time, Mary Brunkow and Fred Ramsdell were working in a company developing drugs for autoimmune diseases. Brunkow and Ramsdell aimed to find the mutated gene causing scurfy, unfortunately, genome mapping was not as accessible as it is today. After years of work, they found the mutation in the FOXP3 gene.
They suspected the human variant of the scurfy mice disease might be IPEX, a rare autoimmune disease. After collecting and analysing the samples from patients with IPEX, they found harmful mutations again in the FOXP3 gene. In 2001, they revealed that mutations in the FOXP3 gene in both humans and mice cause autoimmune diseases. Later, Shimon Sakaguchi and other researchers proved that the FOXP3 gene regulates the development of regulatory T-cells; which prevent the body from attacking itself, important for a process called peripheral immune tolerance. Mary Brunkow, Fred Ramsdell and Shimon Sakaguchi’s discoveries provided vital information about our bodies, which will result in revolutionary medical treatments in the future.

Sources:
https://www.nobelprize.org/prizes/medicine/2025/press-release/

Leave a comment