By Meadhbh Javier Ni Bhroin
What if there was a substance that could pack the surface area of two football fields into a single gram?
This is exactly what earned Richard Robson, Susumu Kitagawa, and Omar Yaghi the 2025 Nobel Prize in Chemistry. These three men have gained worldwide recognition for their work on developing a new class of crystalline material with the potential to tackle some of the most pressing issues facing humanity today – climate change, clean energy, and pollution.

Metal-Organic Frameworks, or MOFs for short, are highly porous, highly modifiable materials through which gases and chemicals can flow. They act almost like a sponge, trapping molecules within their pores. These pores can be customised to carry out specific functions, like harvesting light, or catalysing a reaction.
They are made of secondary building units (SBUs) – clusters of metal ions that are linked together using organic, carbon-based molecules to form a three-dimensional structure. Did you ever play with K’Nex as a kid? Think of SBUs as the connectors and organic linkers as the rods.
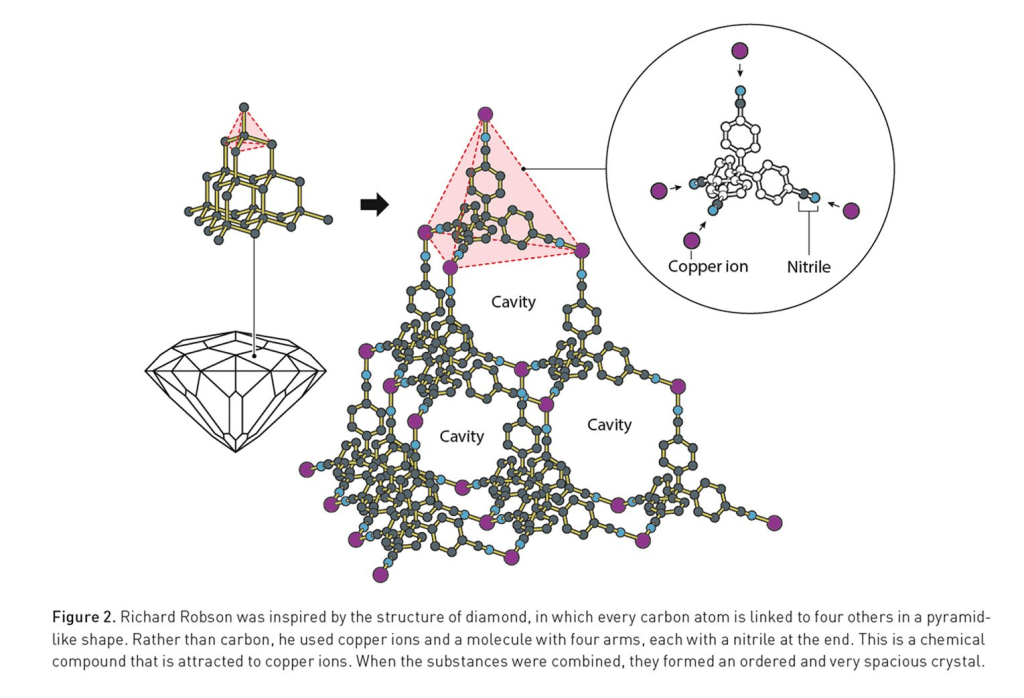
It all started in 1974, when Robson was building models of crystalline structures for chemistry students at the University of Melbourne. ‘As I was constructing these models – putting metal rods of clearly defined dimensions into wooden balls with accurately drilled holes – the thought arose: what if we used molecules in place of the balls and chemical bonds in place of the rods?’ he told Adam Smith on October 8th [1.]. In 1989, he combined positively charged copper ions with a four armed molecule, which allowed for the formation of an organised crystal structure similar to diamond, filled with many, many pores, as stated in the Nobel Prize Press Release [2.]. Although Robson’s creation was unstable, it was the beginning of a new and promising field of chemistry.
Following the discovery, Kitagawa and Yaghi separately carried out crucial research to further develop this field. Kitagawa demonstrated that these materials could capture and release gases, and also predicted the physical and chemical properties of MOFs. Yaghi, on the other hand, managed to create stable structures using multiple clusters of metal ions. He invented the concept of SBUs and is considered the founder of the field of reticular chemistry – linking molecular building blocks with organic linkers.
Due to their high stability, surface area, and modifiability, MOFs have a wide range of potential applications. These include the capture, storage, catalysis of fuels like methane and hydrogen, which are more energy-efficient than fossil fuels and produce cleaner by-products. Much MOF research focuses on capturing carbon dioxide, one of the most abundant greenhouse gases, from the atmosphere. MOFs may also be used to clean wastewater, filtering out organic and inorganic substances. There is even potential for MOFs to be used in biomedical applications, like drug delivery [3.].
However, there are difficulties in producing MOFs at a large scale. They don’t always maintain their stability outside the lab, when exposed to humidity and heat. In addition, the cost of raw materials, solvent, and the long synthesis times are major barriers to production. Even Professor Robson himself is sceptical, telling BBC News: ‘There’s talk about binding CO2 and solving the world’s atmospheric problems, which don’t sound realistic to me – but these sorts of compounds could do that sort of job on a small scale’ [4.].
It is undeniable that this rapidly evolving field shows great promise for science, but for these researchers, it is the culmination of a lifetime of hard work. Kitagawa expressed his hope for these materials during a Nobel news conference: ‘My dream is to capture air and separate air – for instance, CO2 or oxygen or water – and convert this to useful materials using renewable energy.’ [1.]
Yaghi, who grew up in a crowded single-room home in Jordan with a Palestinian refugee family, described science as ‘the greatest equalising force in the world’ in an interview with Adam Smith on October 8th [1.]. His incredible strides in research are no doubt driven by his passion for chemistry, telling Chemistry World in 2019: ‘I’m in love with chemistry. Hobbies are something you do for enjoyment and relaxation – and for me that’s chemistry.’ [1.]
According to the University of Melbourne Newsroom, Robson described winning the award as ‘a great honour and pleasure’, crediting much of his success to his collaborators, Bernard Hoskins and Brendan Abrahams. University of Melbourne’s Prof. Mark Cassidy describes him as ‘a humble man, who has achieved this honour by simply doing what he loves – going into the lab every day, talking to students, thinking big chemistry thoughts for decades and running experiments.’ [5.]
The work these scientists have achieved is a reminder of the power of curiosity, collaboration and perseverance in shaping the future of our world.

Sources:
- https://www.chemistryworld.com/news/meet
the-three-scientists-who-have-just-won-the-nobel-prize-in-chemistry/4022291.article - https://www.nobelprize.org/prizes/chemistry/2025/press-release/
- https://shop.nanografi.com/blog/what-are-the-applications-of-metal-organic-framew
orks - https://www.bbc.com/news/articles/c0r0l742kpjo
- https://www.unimelb.edu.au/newsroom/news/2025/october/professor-richard-robson-wins
-nobel-prize-in-chemistry


Leave a comment