Keep this in mind when getting a chemical formula tattooed!
By Irene Verburg Pérez
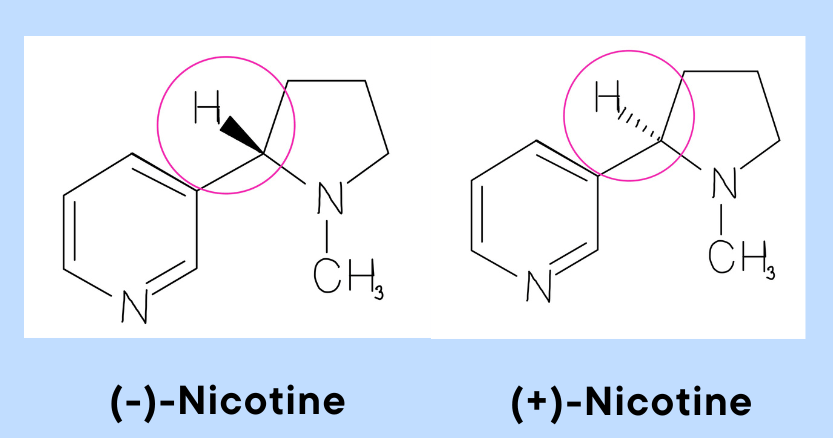
In recent years it has become fairly popular to get a chemical formula tattooed, this is usually a molecule which the individual uses, such as caffeine or nicotine, or common neurotransmitters such as adrenaline, dopamine or serotonin. What people don’t usually know is that many of these have an enantiomer, meaning that the mirror image of the molecule is a different one and may have different activity. For example nicotine, its naturally occurring enantiomer, the one consumed, and therefore the one you may aim to get tattooed is (-)-nicotine. Its (+)-nicotine enantiomer however, is much less psychoactive (and less toxic)! Similarly for adrenaline, its mirror image (D-adrenaline) is about 15-40 times less active! So if you’re thinking of getting a chemical tattoo, make sure you get the right enantiomer!
Lightning, the atmosphere’s cleaning agent
By Inés Martínez Marchán
Lightning produces molecules that can “clean” the atmosphere, such as hydroxyl (OH) and hydroperoxyl (HO2), but also produces amounts much greater than expected due to weaker, subvisible electrical discharges. These subvisible electrical discharges are generated by the collisions between ice particles and water droplets, causing separation of charges: positive charges accumulate at the top, negative charges at the bottom. This generates an electric field between cloud and ground (or even within the cloud) that becomes strong enough to start partial ionization of air, generating these subvisible discharges that take the form of tiny ionized paths in the air.
The previously mentioned molecules are produced in large quantities when Nitrogen, oxygen, and water vapor molecules are broken down due to electric forces. They dominate the atmospheric oxidation chemistry that removes the majority of trace gases, including some greenhouse gases. Mainly, hydroperoxyl acts as a recycler of the hydroxyl radicals, which are the ones in charge of converting the pollutants such as methane and carbon monoxide into less harmful substances like carbon dioxide or water! So next time you see a thunderstorm, take it as a really loud cleaning spree!

Photo by Leon Contreras on Unsplash
Fishing for… pathogens?
By Jacob Whiteman Salmerón
In a war spanning billions of years, parasites and hosts have been in an arms race, trying their best to one-up one another. This has consequently given rise to many ingenious ways of overcoming the opponent; from retreating into an impenetrable spore capsule for decades until conditions are right, to having the ability to create antibodies capable of neutralising every target possible, both sides have been hard at work, quickly countering each other’s newest weapons.
Along the way, however, in its ever-surprising nature, evolution has come up with some more… interesting and creative solutions to the problem of defence.
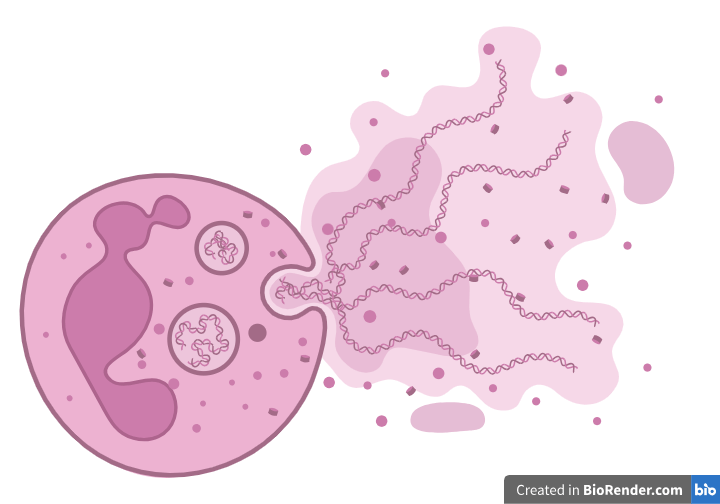
One of these solutions consists of a fishing net-like structure made of the DNA of neutrophils, aptly called “NETs”, or Neutrophil Extracellular Traps. The DNA of neutrophils, one of the main types of white blood cells, becomes imbued with anti-microbial proteins and is spewed out into the surrounding area. The DNA and its accessory molecules make it sticky and toxic to microbes, trapping them in the NET and either killing them slowly or making it easy for other cells to come over and eat them up.
While this does kill the neutrophil (it is the equivalent of us spewing out our brains to catch a swarm of ants), it kills many pathogens at once, and neutrophils are quickly renewing cells, meaning the loss is hardly noticeable. While pathogens have adapted to it by creating compounds that can basically cut through it (like if fish started carrying scissors around to cut fisherman’s nets), it is still an extremely effective method and testament to the immune system’s ingenuity when it comes to fighting our most feared parasites and intruders.
The chemistry behind the perfect smile
By Irene Verburg Pérez
Everybody wants a smile that lights up a room, but how far are you willing to go to get this? Many people use teeth whitening strips. It is a relatively cheap method of getting rid of those stubborn coffee stains, but have you ever wondered how it works? It is simple chemistry!
The strips contain a H2O2 gel that acts on the enamel of the teeth, which is the outermost layer. It is a crystalline semi-porous solid mainly composed of calcium and phosphorous, practically colourless, but as it is porous it is susceptible to staining. Due to the high contact time that the strips require to act, the hydrogen peroxide gel seeps into the pores. As it comes into contact with water it decomposes making oxygen radicals, which are highly reactive oxidising agents. The radicals then break down the large organic compounds that cause the stains into smaller particles that can be washed away.
While this is a great way to make your teeth whiter it also comes with its consequences. As forementioned, the oxygen radicals are very reactive species. They react with anything in their path, like the gums or even with the nerves within the teeth making them have a higher sensitivity to certain foods and temperatures. Not only this, but they can also have an uneven finish to them, being whiter in some parts of the teeth than others.
Overall, while teeth whitening strips can be effective to boost confidence with a killer smile, it is important to be aware of the risks of radical chemistry.

The sounds of terror
By Inés Martínez Marchán
You would be surprised by the extent to which physics is related to the audiovisual arts, especially in horror movies, as one of its key components is the use of sound and its effect on the human body.
In horror movies, due to their very different physical properties, two distinct types of waves can be distinguished: high-frequency waves and low-frequency waves. High-frequency waves around 2-10 kHz propagate sharply, rapidly changing energy that the ear detects quickly, making them ideal for jump scares. An instantaneous alarm is triggered in the body caused by the sudden detection of the sound wave, and due to the fact that high-frequency sounds are harder for our ears to localize, it is a really powerful tool to create unease.
On the other hand, low-frequency waves, especially those below 20 Hz, create fear because their large wavelengths can pass through walls, vibrate objects, and resonate with the human body, producing discomfort or anxiety even when the sound is barely audible. This is often used to build tension before a climax and explains the usual feeling of paranoia before an important scene.
Lastly, another wave characteristic used in the production of horror movies is the use of distorted or nonlinear sounds. Distorted or non-linear sounds are harsh, irregular harmonic structures that the auditory system struggles to decode because the resultant oscillations don’t match typical natural patterns.
These physics-based manipulations of wavelength and resonance form the foundation of how horror movies use sound to produce fear, building on how physics is present in our everyday lives. If you are one of those people who try to rationalise horror movies to not be scared, we hope the curiosity corner was able to give you an additional argument!

Photo by Dr. Keith Bound in LinkedIn

Leave a comment