By Maria Del Socorro
What do you imagine when you think of fireworks? Maybe New Year’s Eve, or a crowded stadium. Maybe protests in Georgia or your local Tesco at 2AM. A violent display symbolic of a fresh start, a rebellion, a break in the ordinary of everyday life. A massive spectacle meant to reflect intimate wishes for a new life, maybe even a new you. So how did these multi-colour flares come to be, and what’s behind the art of bringing metal and fire to life?
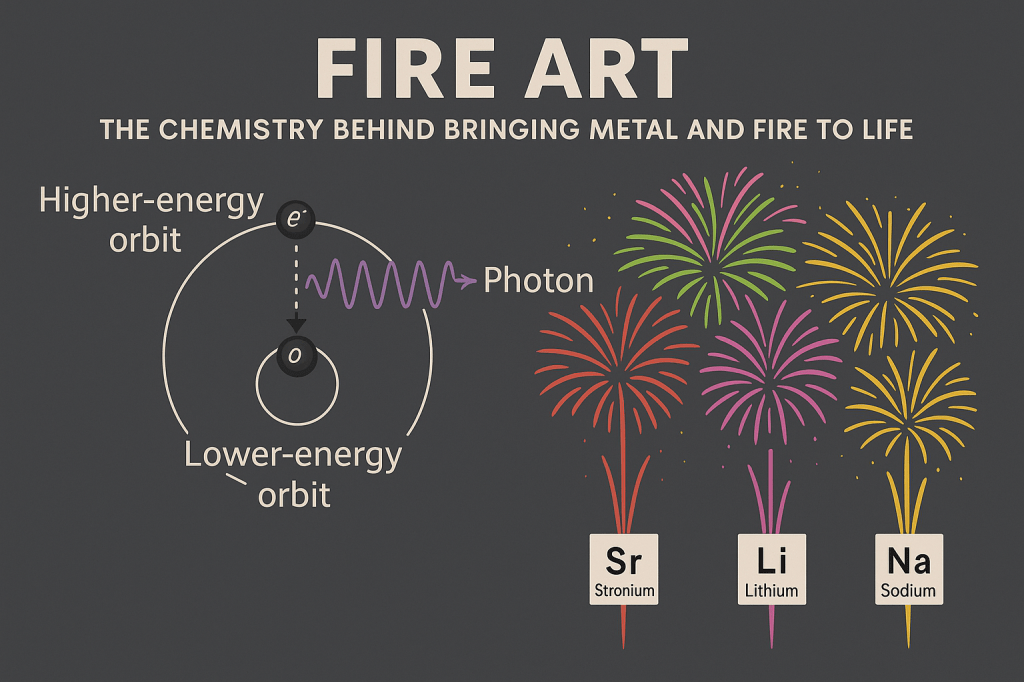
Image created with AI
The earliest records of firecrackers date back to 200 B.C.E in Liuyang, China, according to the American Pyrotechnics Association (1). People would ward off spirits by throwing hollowed out bamboo onto fires, creating a loud bang when they burst. Some hundred years later, Tang dynasty alchemists mixed nitrate salts, sulphur and charcoal, leading to the discovery of gunpowder. There are three tenets to fire: oxygen, heat, and fuel. Usually oxygen is provided through the air, but in gun powder the nitrate salts themselves (ie. KNO3) act as the source.
Charcoal provides the fuel; its porous nature makes it especially effective as the nitrate salts and sulphur can sit far within the charcoal. Sulfur brings down the activation energy of the reaction, so that less heat is required for spontaneous ignition. If the activation energy is too large, the reaction will either take a long time to take place or won’t occur at all. It takes a lot of heat before potassium nitrate and charcoal can spontaneously ignite. The reaction which sulphur undergoes is difficult to elucidate but it is known that it is exothermic, meaning it releases heat. The reaction also occurs at much lower temperatures than potassium nitrates ignition point, leading to a “preheating” effect making ignition faster according to Andy Brunning, Compound Interest (2).
The golden ratio for this explosive concoction is roughly 75% potassium nitrate, 10% sulphur, and 15% charcoal. For any curious readers, yes all these reagents can be found in Cocker labs ;).
However, if you were to throw these reactants in a bowl and light them, you would probably get a cute fire at best. To create a firework you need confined space. This brings all the reactants much closer, forcing them to react faster and more often, creating the pressure that gives the iconic explosion. For firework displays, the confined space is provided by aerial shells. Shells generally contain black powder, stars (spheres), and a timed fuse down the middle. Typically, at the base of the shell there will also be a lift charge. This compartment is filled with black powder which sends the fireworks upward. The orientation of the stars within the shell is an evolving art. Different configurations and ratios of the stars create different firework shapes or effects.
Now that we know how to get them up and exploding in the air, we need to bring in the colours, which will require some quantum mechanics. Each “star” will contain a metal element which is heated during the combustion process. Thermal energy from this is absorbed by the metal’s electrons. Remember, electrons can only exist at certain energy levels around the nucleus, the same way we can only stand on the rungs of a ladder. With the absorbed energy, the electron moves to a higher level, known as the excited state. However, it can’t stay there forever and as the electron goes back down to its ground state, it releases energy specific to the gap between these two levels. The released energy is known as a photon – which is essentially a package of light. For us this is perfect because where there is light there is colour, and certain elements have transitions exactly suited to create these colours.
Fireworks are an ancient art but nonetheless have an evolving future. Modern software allows for finely tuned design of a fireworks show. New materials and aerial shells allow for never-before-seen displays. Environmental impacts are also changing the nature of the industry. Companies must innovate new fireworks that leave a mark on the audience without doing the same to the atmosphere. Many developments have also been made in low-noise fireworks, designed so that the whole family – pets included – can enjoy. Thus, whilst much development has been made to create these incredible spectacles, much is yet to come for this beloved fire art.
References
- History of Fireworks, https://www.americanpyro.com/history-of-fireworks, (accessed
October 26, 2025). - A. Brunning, The Chemistry of Gunpowder,
https://www.compoundchem.com/2014/07/02/the-chemistry-of-gunpowder/, (accessed
November 22, 2025). - Veritasium, The Hidden Science of Fireworks,
https://www.youtube.com/watch?v=lfkjm2YRG-Q, (accessed October 26, 2025).

Leave a comment